An impactful winter storm is possible next week, so you may want to prepare for it. Most people are familiar with how their driveways and sidewalks look like this every winter. But how does this salt help clean roads?
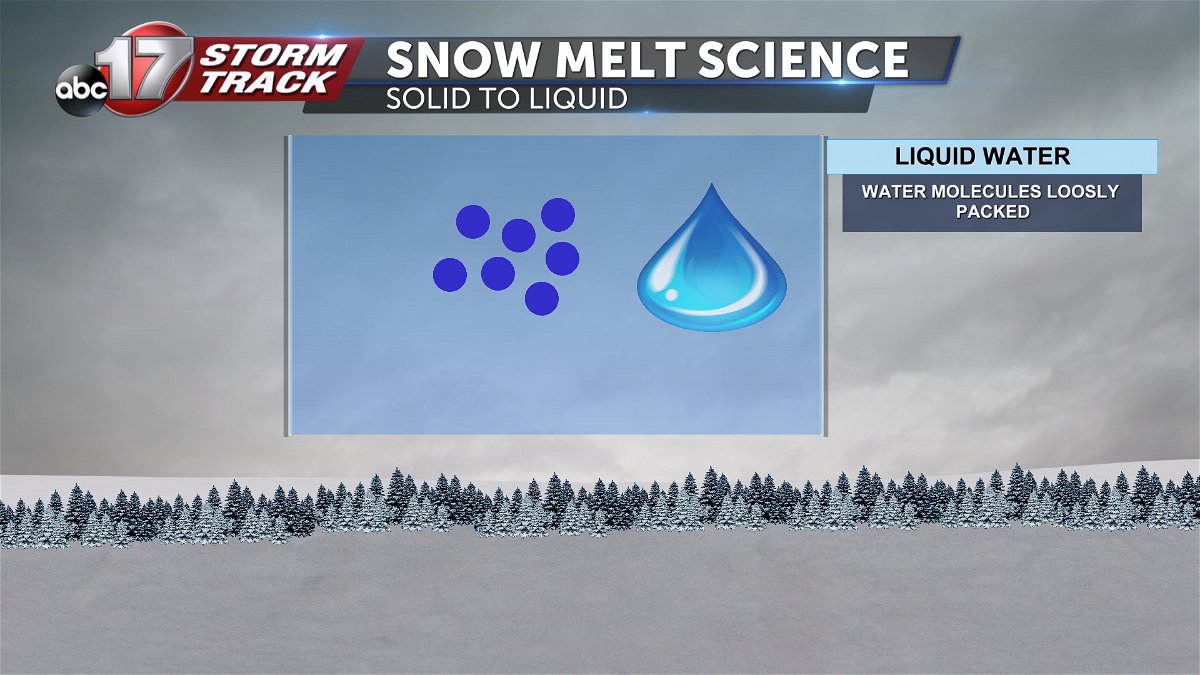
Water in its liquid state is made up of loosely packed molecules. This allows the liquid to flow easily and conform to the shape of the container.

When water reaches its freezing point of 32 degrees or below, the molecules become more closely spaced, giving it a solid shape.
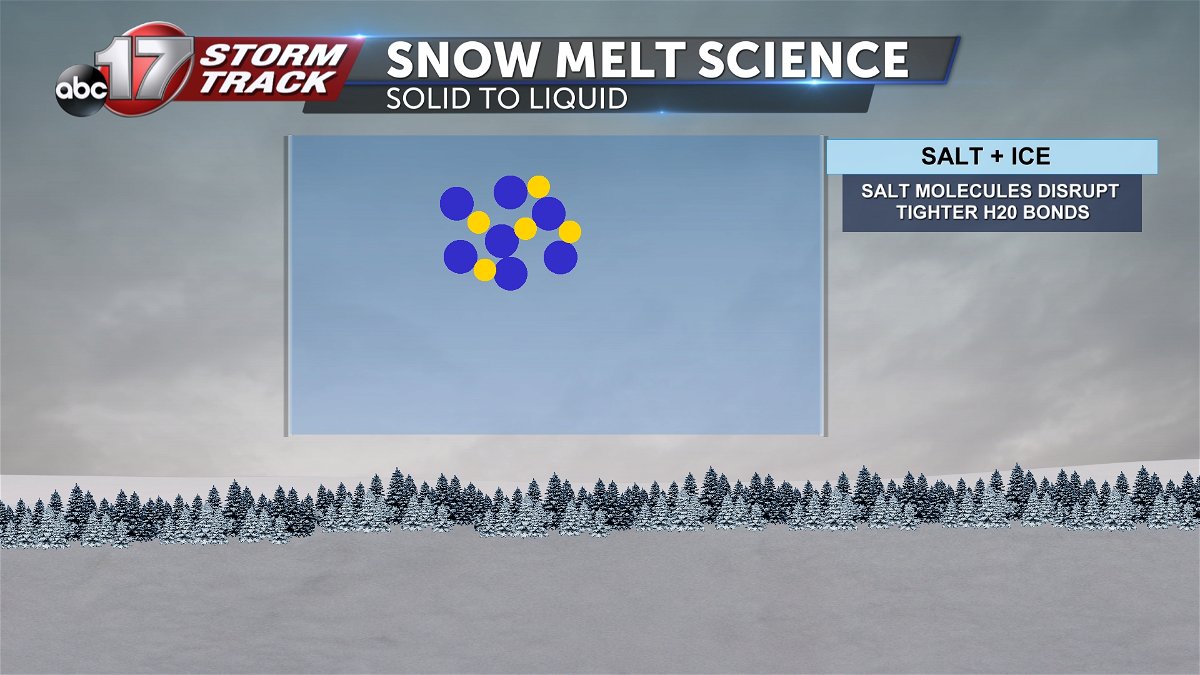
Salt does not necessarily melt ice or snow due to increased heat. It simply lowers the freezing point of water and lowers its temperature. At the molecular level, salt molecules interfere with the bonding of h20 molecules, returning water to its liquid state.

As temperatures drop, it’s also important to understand what ingredients are in your salt. The effectiveness of a salt at temperatures well below freezing is determined by the composition of the compound.
Also, using a water mixture can be more cost-effective as less brine is used and more areas can be fused. The downside is that the freezing temperature increases, so be careful if the temperature is cold. It’s also important to avoid consuming too much salt, which can harm the environment, and to always check the pet safety seal to keep your furry friend healthy.
ABC 17 News is committed to providing a forum for civil and constructive conversation.
We ask you to keep your comments respectful and relevant. Click here to review our Community Guidelines.
If you would like to share your story idea, please submit it here.
