The proposed composite membrane meets the U.S. Department of Energy’s highly challenging technology goal of use in automotive fuel cells by 2025, paving the way for powerful and affordable electric vehicles.Credit: Kenji Miyatake, Waseda University and Yamanashi University
× close
The proposed composite membrane meets the U.S. Department of Energy’s highly challenging technology goal of use in automotive fuel cells by 2025, paving the way for powerful and affordable electric vehicles.Credit: Kenji Miyatake, Waseda University and Yamanashi University
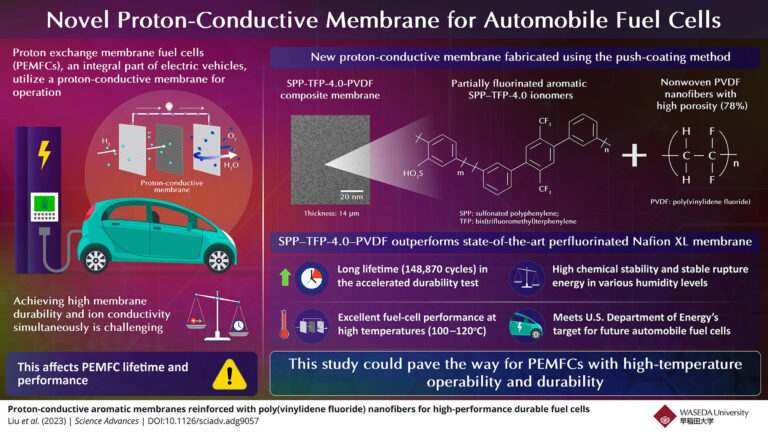
A fuel cell is a compact energy conversion unit that takes a clean energy source, such as hydrogen, and converts it into electricity through a series of redox reactions. Specifically, polymer electrolyte fuel cells (PEMFCs), which are an integral part of electric vehicles, utilize proton-conducting membranes for operation. Unfortunately, these membranes have a trade-off between high durability and high ionic conductivity, which affects the lifetime and performance of PEMFCs.
To overcome this problem, scientists have synthesized chemically and physically modified perfluorosulfonic acid polymer membranes such as Nafion HP, Nafion XL, and Gore-Select. These have proven to be much more durable than the unmodified membranes traditionally used in fuel cell operation.
Unfortunately, all existing proton-conducting membranes have difficulty passing accelerated durability tests or combined chemical and mechanical tests set by the U.S. Department of Energy (DOE) to facilitate their use in automotive fuel cells. does not meet difficult technical goals. 2025.
Now, a group of Japanese researchers led by Professor Kenji Miyatake from Waseda University and the University of Yamanashi have synthesized a new proton-conducting membrane for PEMFC.Their works published in magazines scientific progressis co-authored by Dr. Liu Fanghua of Waseda University and Yamanashi University and Dr. Ick Soo Kim of Shinshu University.
The researchers developed a polymer made of a partially fluorinated aromatic ionomer (thermoplastic resin stabilized by ionic crosslinking) called SPP-TFP-4.0 (SPP: sulfonated polyphenylene, TFP: bis(trifluoromethyl)terphenylene). A proton-conducting membrane was synthesized using molecular materials.
Next, a push-coating method was used to coat electrospun nonwoven isotropic poly(vinylidene fluoride) (PVDF) nanofibers with high porosity (78%) or porous expanded polytetrafluoroethylene (ePTFE). ) was used to strengthen the ionomer. This resulted in composite membranes, SPP-TFP-4.0-PVDF and SPP-TFP-4.0-ePTFE, with thicknesses of 14 μm and 16 μm, respectively.
The researchers performed various tests on these proton-conducting membranes and demonstrated that the PVDF-reinforced membranes were superior. “We surpassed state-of-the-art chemically stabilized and physically reinforced perfluorinated Nafion XL membranes in terms of fuel cell operation and in-situ chemical stability at high temperatures of 120°C.”ahThe temperature is low at ℃ and the relative humidity is 30%,” Miyatake said.
The SPP-TFP-4.0-PVDF membrane demonstrated a long life of 148,870 cycles or 703 hours in an accelerated durability test with frequent dry-wet cycles under open-circuit voltage conditions. This is more than seven times the DOE goal. Additionally, it exhibited high chemical stability with little degradation, stable energy to fracture at different humidity levels, and very stable mechanical properties from 0 to 60% relative humidity at 80 °C.ahExcellent fuel cell performance at high temperatures (100-120°C)ahC).
Indeed, the proposed aromatic polymer-based reinforced proton-conducting membrane meets the US Department of Energy’s goals for future automotive fuel cells, making it an advantageous alternative. Therefore, this study may pave the way for his PEMFCs with high temperature operability and durability. “As a result, fuel cell-based electric vehicles could become more powerful and affordable. This would also contribute to the realization of a hydrogen-based, decarbonized society,” concludes Miyatake. .
For more information:
Fanghua Liu et al. Proton-conducting aromatic membranes reinforced with poly(vinylidene fluoride) nanofibers for high-performance durable fuel cells, scientific progress (2023). DOI: 10.1126/sciadv.adg9057
Magazine information:
scientific progress